Since the noble gases are a special group because of their lack of reactivity, the element fluorine is the most reactive nonmetal. The most reactive nonmetals reside in the upper right portion of the periodic table. The next two members are oxygen and chlorine.Fluorine, chlorine and bromine are. Due to their highly reactive nature, these elements are commonly stored as an inert fluid material, such as petroleum-derived oil. Nonmetals tend to gain electrons in chemical reactions, and have a high attraction for electrons within a compound. The most reactive metals are located at the left top of the Periodic Table. Of all the elements on the periodic table, reactive nonmetals have the highest ionization energy and electronegativity, making them likely to form ionic bonds with metals of very low ionization. The halogens, periodic table column 17, are the most reactive nonmetals, because they have the highest electronegativities and therefore can strongly attract electrons from almost any other. Solution Non-metal in the periodic table: The non-metals are the elements that are located on the right side of the periodic table. Metals are found on the left and in the middle, whereas. Non-metals can be gases, liquids or solids. The periodic table can be used to find out if an element is a metal or a non-metal. 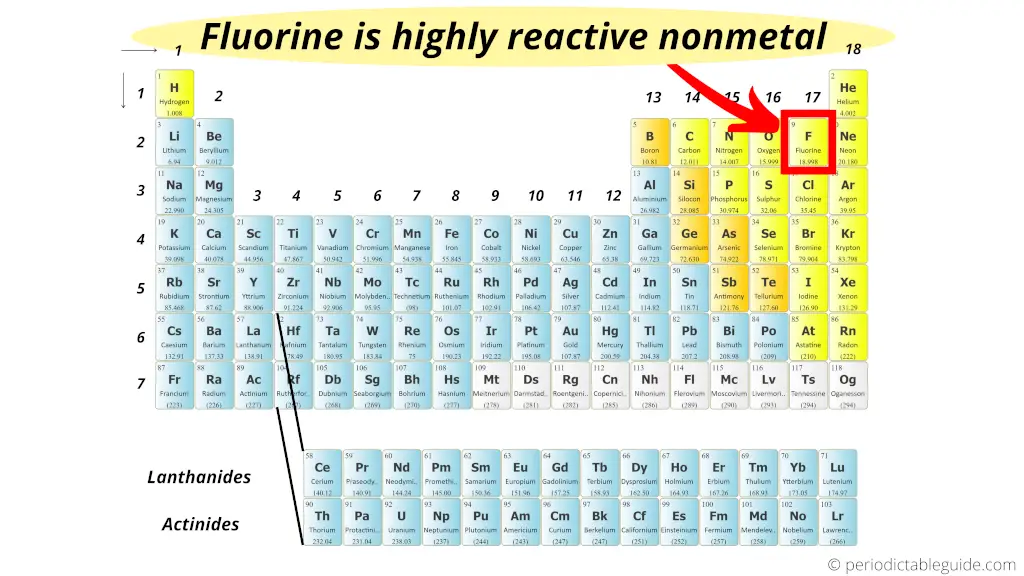
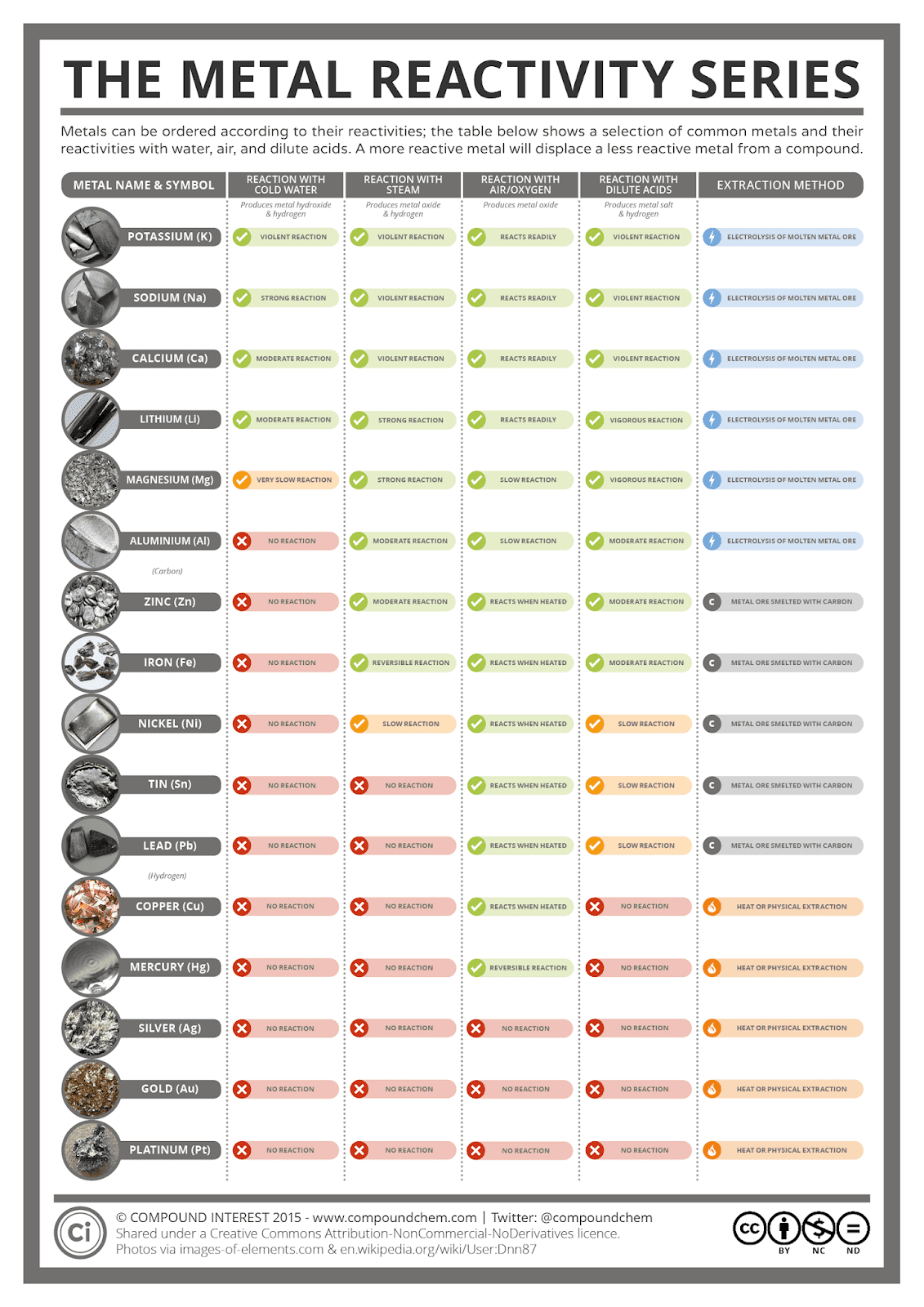
In general, the more reactive a metal is: This table summarises the reactions of some metals in the. They are non-lustrous, brittle and poor conductors of heat and electricity (except graphite). The reactivity series of metals is a chart listing metals in order of decreasing reactivity. periodic table may be classified into three categories: metals, located on the. These are electronegative elements with high ionization energies. most common electronegativity scale is the Pauling electronegativity scale. With respect to the reactivity of metallic elements listed on the periodic table, the metallic elements are broadly classified into four groups:Īctive metals are characterized by their tendency to readily combine with gaseous oxygen and atmospheric water vapor due to a single electron in its outer shell that can be readily exchanged to form a cation and ultimately lead to a chemical reaction. Elements that tend to gain electrons to form anions during chemical reactions are called non-metals. These elements belong to groups IA and IIA of the periodic table. 
The most active metals in the activity series are lithium, sodium, rubidium, potassium, cesium, calcium, strontium and barium. very reactive reactive lose electrons in chemical reactions On the periodic table: Identify and name the metals (shown in pink), the non-metals (green). Trending Questions What actually transferred into the water? Subatomic particles in chemical reactions? Why is a melting ionic compound a better conductor than a solid ionic compound? Are glow sticks an example of a combustion reaction? What is the strongest intermolecular force between molecules of ch3ch2oh? How would you describe acids with regards to their reaction to litmus and gumamela extract? How do you make more hydrogen from water? How does potassium -39 different from potassium -41? Is photolysis the splitting up of water into hydrogen and oxygen? What is the scientific name for carbonated water? 4.All active metals are found in Group I of the periodic table (i.e., on the left side of the periodic table), except hydrogen, which is not considered a metal and lies at the top left hand corner of the periodic table. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
